- 路 Microwave
- 路 Atmospheric Pressure Microwave 路 Pressure Microwave 路 Parallel Microwave
- 路 Ultrasonic 路Low Temperature Ultrasound
- 路 Ultraviolet Light
- 路 Microwave Heating 路 Atmospheric Pressure Synthesis 路 Atmospheric Pressure Catalysis 路 Atmospheric Pressure Extraction
- 路 Sample Preparation 路 Microwave Digestion
- 路 Soil Digestion 路 High Pressure Synthesis
- 路 Solid Phase Synthesis
- 路 Organic Synthesis
- 路 Ionic Liquid Synthesis
- 路 Degradation Of Natural Organic Matter
- 路 Natural Product Extraction / Purification
河北祥鹄科学仪器有限公司
300B Complex of Cu_Sm_Cu trimetals _ omitted __1_2_thiophene_ethyl_1H_pyrrole_
This research was completed by a researcher at the School of Chemical and Pharmaceutical Sciences of Jingchu Institute of Technology to discuss the catalytic synthesis of 2-ethyl-5-[2-nitro-1-(2-thiophene) by the complex of Cu-Sm-Cu trimetallic. The paper of ethyl-1H-pyrrole was published in the important journal
Eu3 + doped LaF3 and CaF2 phosphors with special fluorescence properties were successfully synthesized by microwave method. The structure, morphology and luminescence properties of the samples were characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM) and photoluminescence spectroscopy (PL). The effects of sodium tartrate and Eu3 + doping on the luminescence properties of synthetic phosphors were discussed. The experimental results show that the addition of sodium tartrate and the doping amount of Eu3 + have a great influence on the fluorescence performance. The change of molar ratio makes Eu3 + change from magnetic dipole transition to electric dipole transition dominated, and then the main peak The position changes from the previous 588 nm to 613 nm. However, it did not cause changes in other peak positions, indicating that Eu3 + entered the lattice of LaF3 and CaF2 without any other changes.

Fig.1/3↑
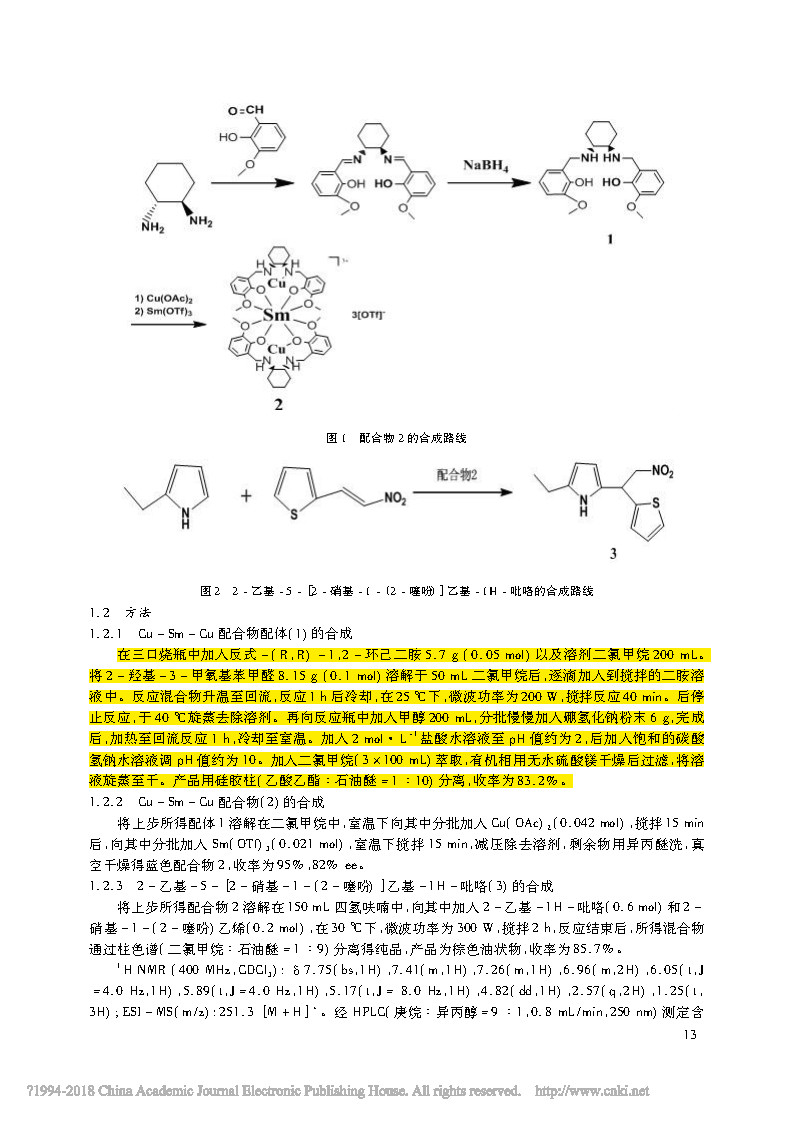
Fig.2/3↑

Fig.3/3↑
The Cu-Sm-Cu complex 2 is synthesized from trans-(RR)-1,2-cyclohexanediamine and 2-hydroxy-3-methoxybenzaldehyde, and the complex is used as a catalyst. 2-Ethyl-5-[2-nitro-1-(2-thiophene)]ethyl-1H-pyrrole was catalyzed under microwave conditions. Compared with the methods reported in the literature, it has the characteristics of high yield and short time, mild reaction conditions and environmental friendliness. At the same time, the effects of reaction solvent, reaction time and microwave power on the reaction yield of ligand 1 were investigated. The catalyst has the advantages of good selectivity, simple operation and high yield.
In a three-necked flask, 5.7 g (0.05 mol) of trans-(R,R)-1,2-cyclohexanediamine and 200 mL of a solvent dichloromethane were added. After dissolving 2-hydroxy-3-methoxybenzaldehyde 8.15 g (0.1 mol) in 50 mL of dichloromethane, it was added dropwise to the stirred diamine solution. The reaction mixture was warmed to reflux, and the reaction was cooled for 1 h, and the microwave power was 200 W at 25 ° C, and the reaction was stirred for 40 min. After the reaction was stopped, the solvent was removed by rotary evaporation at 40 °C. Further, 200 mL of methanol was added to the reaction flask, and 6 g of sodium borohydride powder was slowly added in portions, and after completion, it was heated to reflux for 1 hour, and cooled to room temperature. Add 2 mol · L - 1 aqueous hydrochloric acid to a pH of about 2, then add a saturated aqueous solution of sodium bicarbonate to adjust the pH to about 10. Dichloromethane (3 × 100 mL) was added for extraction. The organic phase was dried over anhydrous magnesium sulfate and filtered and evaporated. The product was isolated on a silica gel column (ethyl acetate: petroleum ether = 1 : 10).

















































 京ICP备15050585号
京ICP备15050585号

