- 路 Microwave
- 路 Atmospheric Pressure Microwave 路 Pressure Microwave 路 Parallel Microwave
- 路 Ultrasonic 路Low Temperature Ultrasound
- 路 Ultraviolet Light
- 路 Microwave Heating 路 Atmospheric Pressure Synthesis 路 Atmospheric Pressure Catalysis 路 Atmospheric Pressure Extraction
- 路 Sample Preparation 路 Microwave Digestion
- 路 Soil Digestion 路 High Pressure Synthesis
- 路 Solid Phase Synthesis
- 路 Organic Synthesis
- 路 Ionic Liquid Synthesis
- 路 Degradation Of Natural Organic Matter
- 路 Natural Product Extraction / Purification
河北祥鹄科学仪器有限公司
800C Dissolution conditions of waste nickel-hydrogen battery in citric acid_Preparation of _0_5_Fe_2O_4
This research by the researcher of the College of Chemistry and Chemical Engineering of Henan Normal University, discusses the dissolution conditions of waste nickel-metal hydride batteries in citric acid and the research on the preparation of Ni0.5Co0.5Fe2O4, published in the important journal
The dissolution conditions of the cathode material of the waste nickel-hydrogen battery in citric acid were studied. The effects of initial concentration of citric acid, liquid-solid ratio, dissolution temperature and dissolution time on the dissolution rate of cobalt and nickel were analyzed by single factor and orthogonal test. The experimental results show that when the concentration of citric acid is 2 mol/L, the liquid-solid ratio (mass ratio) is 8:1, the dissolution temperature is 90 °C, and the dissolution time is 50 min, the dissolution rate of nickel-cobalt is the highest. Under the optimal dissolution conditions, the spherical nano-scale Ni0.5Co0.5Fe2O4 with good performance was prepared by microwave hydrothermal method. The magnetic parameters were: saturation magnetization 50.312 A · m2/kg, residual magnetization 15.306 A· m2 /kg, coercivity is 0.052726 T.

Fig.1/3↑

Fig.2/3↑
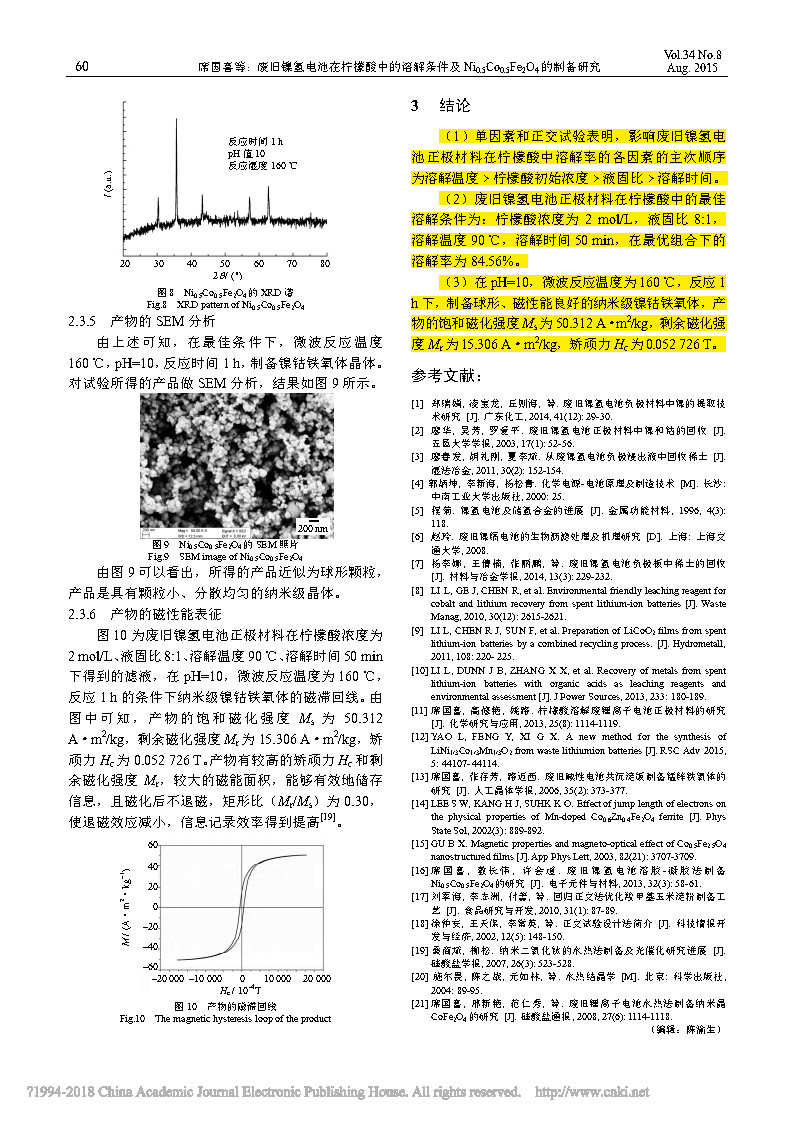
Fig.3/3↑
(1) Single factor and orthogonal tests show that the main order of factors affecting the dissolution rate of the cathode material of waste nickel-hydrogen battery in citric acid is dissolution temperature > initial concentration of citric acid > liquid-solid ratio > dissolution time. (2) The best dissolution conditions of the spent nickel-hydrogen battery cathode material in citric acid are: citric acid concentration of 2 mol/L, liquid-solid ratio of 8:1, dissolution temperature of 90°C, dissolution time of 50 min, in the optimal combination The lower dissolution rate is 84.56%. (3) Nano-scale nickel-cobalt ferrite with good spherical and magnetic properties was prepared at pH=10, microwave reaction temperature of 160 °C and reaction for 1 h. The saturation magnetization Ms of the product was 50.312 A · m2/kg, residual magnetization. The strength Mr was 15.306 A· m2/kg, and the coercive force Hc was 0.052726 T.
The content of Ni2+, Co2+ and Fe2+ in the filtrate was determined by atomic absorption spectrophotometer, and then the appropriate amount of nickel sulfate and cobalt sulfate was added to the filtrate according to the molar ratio r(Ni2+:Co2+:Fe2+)=1:1:4[16]. And ferrous sulfate, heated in a 60 °C water bath, add 2 mol / L sodium hydroxide under stirring, adjust pH = 8 ~ 12, boil for 1 h, microwave digestion at 120 ~ 160 ° C for 0.5 ~ 2 h, cooling Filter, wash, and transfer the product to an electric blast oven (100 ° C) for 1 h. Naturally cooled and ground to obtain a nickel-cobalt ferrite powder.

















































 京ICP备15050585号
京ICP备15050585号

