- 路 Microwave
- 路 Atmospheric Pressure Microwave 路 Pressure Microwave 路 Parallel Microwave
- 路 Ultrasonic 路Low Temperature Ultrasound
- 路 Ultraviolet Light
- 路 Microwave Heating 路 Atmospheric Pressure Synthesis 路 Atmospheric Pressure Catalysis 路 Atmospheric Pressure Extraction
- 路 Sample Preparation 路 Microwave Digestion
- 路 Soil Digestion 路 High Pressure Synthesis
- 路 Solid Phase Synthesis
- 路 Organic Synthesis
- 路 Ionic Liquid Synthesis
- 路 Degradation Of Natural Organic Matter
- 路 Natural Product Extraction / Purification
河北祥鹄科学仪器有限公司
Preparation of Gd doped carbon Quantum Dots High performance Magnetic Resonance fluorescence double Mode Molecular Image probe b
This paper, by a researcher from the School of Materials Science and Engineering, Southwest University of Science and Technology, discusses the preparation of Gd doped carbon quantum dot-type high performance magnetic resonance fluorescence bimodal molecular imaging probes by microwave hydrothermal method. Published in the Journal of luminescence, an important influential journal in China.
In recent years, the research work of microwave chemical instrument used in the synthesis of materials has become a hot direction of scientific research, which has been paid great attention to by many scholars!
Using both gadolinium and carbon sources as precursors, microwave was used to agitate at molecular level and reach low temperature. The purpose of preparing homogeneous small grain Gd3+ doped carbon quantum dots (Gd3+/ CQDs-MH) in a short time. When the precursor reacts with 45min at 250 ℃, the obtained Gd3+/ CQDs-MH exhibits high quantum yield and strong magnetic resonance properties. It avoids the contradiction that it is difficult to improve the luminescence ability and relaxation property of carbon quantum dots by traditional heating method.
Under this condition, carbon quantum dots of about 1.0nm size have been synthesized. The fluorescence quantum yield is 11.0 and the doping mass fraction of Gd3+ is 16.9.The longitudinal relaxation property is 4545.3 L/mol s ([Gd3+] = 0.01mmol/L). Moreover, the carbon quantum dots have no obvious toxicity to HeLa cells, so it is expected to be used as a high relaxation and high luminescent dual-mode magnetic resonance fluorescence probe.
Microwave hydrothermal method, which uses microwave as heating tool, can be stirred at molecular level, and the heating rate is fast and uniform. Compared with the conventional high temperature pyrolysis method, microwave hydrothermal method has the advantages of low reaction temperature, difficult agglomeration of particles, high purity of carbon quantum dots and narrow size distribution.
Gd3+ with 7 unpaired electrons can reduce the relaxation time of water protons in the tissue and exhibit an enhanced signal. Gd3+ has been detoxified by chelation of polydentate ligands and has been widely used in clinical magnetic resonance imaging contrast agents (Magenville). Carbon quantum dots have excellent fluorescence properties, are non-toxic, and are simple to prepare. Therefore, the magnetic resonance-fluorescence bimodal molecular imaging probe prepared by combining carbon quantum dots with Gd3+ can provide high-resolution structural histological information and achieve highly sensitive functional imaging, overcoming single molecular imaging. The limitations of the technology make the obtained diagnostic results more accurate, which greatly broadens the research scope and application prospects of molecular imaging technology.
In order to avoid the defects of Gd3+/ CQDs prepared by conventional heating method, in which the quantum yield increases and the concentration of Gd3+ decreases slightly, a high concentration of Gd3+ doped carbon quantum dots with high luminescence efficiency is prepared by microwave hydrothermal method. The carbon quantum dots are expected to be used as high performance magnetic resonance fluorescence double mode molecular imaging probes.Document[6] has been proved that increasing the carbonization degree of (GdPM) can increase the content of sp2 hybrid carbon atoms in the carbon nucleus and thus increase the luminescence ability of the carbon quantum dots.
However, the structure of Gd3+—N—O chelating ring in GdPM can be broken at too high pyrolysis temperature, resulting in the loss of Gd3+. Due to the low reaction temperature of microwave hydrothermal method, carbon quantum dots with high Gd3+ doping and high quantum yield can be obtained. In addition, another advantage of hydrothermal method is that the size of the prepared nanoparticles is small and the dispersion is good, which is conducive to the improvement of the luminescence efficiency of carbon quantum dots.

Fig.a↑
(a)The TEM photographs of Gd3+/CQDs-MH prepared by hydrothermal reaction of 45min with microwave are illustrated by electron diffraction patterns and high resolution TEM photographs.
However, the structure of Gd3+—N—O chelating ring in GdPM will be broken at a high pyrolysis temperature, resulting in Gd3+ loss. Due to the low reaction temperature of microwave hydrothermal method, carbon quantum dots with high Gd3+ doping and high quantum yield can be obtained. In addition, another advantage of hydrothermal method is that the size of the prepared nanoparticles is small and the dispersion is good, which is conducive to the improvement of the luminescence efficiency of carbon quantum dots.

Fig.b↑
FTIR Spectra of Gd3+/CQDs-MH and GdPM prepared by Microwave hydrothermal reaction(1:GdPM;2~4:15,30,45min)
Carbon quantum dots with high MRI contrast intensity and high luminescence efficiency were obtained by microwave hydrothermal method using GdPM as the precursor of carbon and gadolinium sources. It is found that microwave hydrothermal method does exhibit the advantages of low temperature and rapid preparation of carbon quantum dots with both high longitudinal relaxation rate and high quantum yield.
At 250 ℃, the quantum yield is 11.0% and the longitudinal relaxation rate is up to 4545.3m·L/mol·s Gd3+/CQDs-MH after only 45 min.However, when the GdPM powder is directly heated in a tube furnace, only when the pyrolysis temperature is high enough (for example, 400 ℃) and the heat preservation is 2 h, can the quantum yield of carbon quantum dots (12.5%) equal to that of Gd3+/ CQDs-MH can be obtained.
However, there is no chelating Gd3+ on the surface of the carbon quantum dots, which can not be used as a MRI- fluorescence bimodal molecular imaging probe because of the loss of magnetic resonance response. When the concentration of Gd3 was low (0.01mmol·L-1), Gd3+/CQDs-MH showed greater relaxation response than Margenvir, which could greatly reduce the required dose of clinical "anchoring" lesions. Because Gd3+ was firmly chelated by CQDs with biocompatibility, Gd3+/CQDs-MH showed no obvious toxicity to cells.
Application of Xianghu instrument in this thesis
GdPM was carbonized by microwave hydrothermal method. Approximately 0.1gGdPM was dissolved in 30mL deionized water and pH value (pH1) was measured. The solution was heated to a certain temperature and kept for a period of time in a microwave hydrothermal parallel synthesizer (XH-800, Beijing Xianghu Science and Technology Development Co., Ltd.). The pressure of the system was recorded (Table 1). After natural cooling to room temperature, a yellowish transparent solution was obtained and the pH value (pH2) of the liquid was measured.

Table 1↑
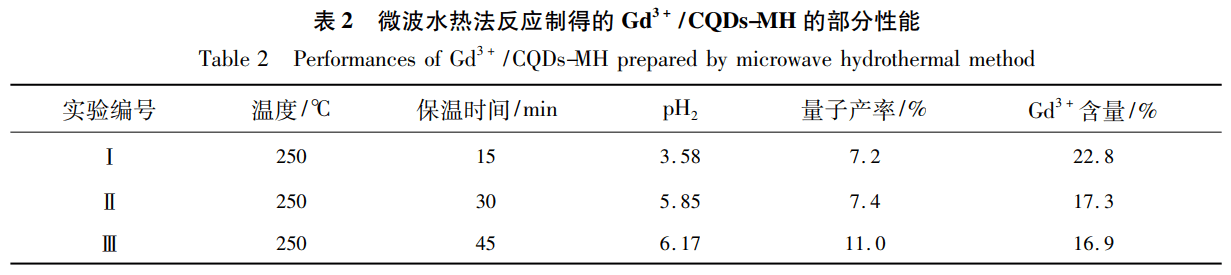
Table 2↑
FTIR Spectra of Gd3+/CQDs-MH and GdPM prepared by Microwave hydrothermal reaction(1:GdPM;2~4:15,30,45min)
Note: XH-800S good performance, the highest temperature control temperature 260 ±0.1 ℃, the highest pressure control pressure 6.0Mpa. this experiment is safe and efficient, lay a solid foundation for the experimental results!

















































 京ICP备15050585号
京ICP备15050585号

